Analysis of Prospective Primary School Teachers’ Knowledge Regarding Chemical Representations on Crystallization Experiment
Abstract
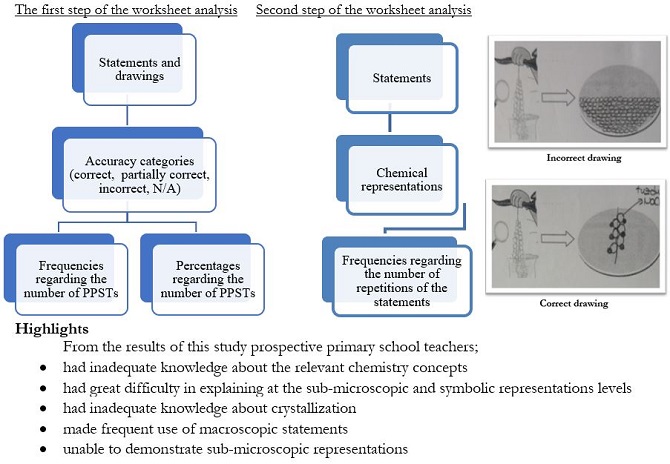
The study aims to determine the subject matter knowledge of Prospective Primary School Teachers (PPSTs) and analyze their chemical representation levels on crystallization. The study was carried out with descriptive research with a qualitative approach based on this purpose. The study participants were eighty freshman students studying at the Department of Primary Education in a state university in the Aegean Region of Turkey. The data were collected using a worksheet containing seven open-ended questions regarding the crystallization experiment. The questions were about the solubility of salt in water, the formation of the salt water and its solubility-temperature graph, the formation of the saturated salt water, and the appearance of particles formed during crystallization. A worksheet was prepared to determine the chemical representation levels of the PPSTs, and the data were subjected to document analysis. The researchers conducted a demonstration experiment and an animated video on the extraction of table salt by crystallization as an activity during the study process. The results indicated that PPSTs' responses related to crystallization were mainly at the macroscopic level. At the same time, they had great difficulty explaining at the levels of sub-microscopic and symbolic representations.
Moreover, they could not explain the concept of dissolution with scientific expressions and distinguish the mixtures from each other. Additionally, most prospective teachers could not draw the correct solubility-temperature graph, so they had difficulty in symbolic representations. The study results imply that to raise the quality of science education in Turkey, PPSTs must attend a quality teaching of science, so primary school students acquire scientifically accurate knowledge of the basic science subjects and concepts such as dissolution, solubility, and crystallization.
Full Text:
Download PDFReferences
Aubrey, C., Ghent, K., & Kanira, E. (2012). Enhancing thinking skills in early childhood. International Journal of Early Years Education, 20(4), 332-348.
Adadan, E. (2014). Investigating the influence of pre-service chemistry teachers' understanding of the particle nature of matter on their conceptual understandings of solution chemistry. Chemistry Education Research and Practice, 15(2), 219-238. https://dx.doi.org/10.1039/C4RP00002A
Akkuzu Güven, N., & Uyulgan, M. A. (2021). Linking the representation levels to a physical separation and purification method in chemistry: Understanding of distillation experiment. Journal of Pedagogical Research, 5 (3), 80-104. https://doi.org/10.33902/JPR.2021370703
Aydeniz, M., Bilican, K., & Kirbulut, Z. D. (2017). Exploring pre-service elementary science teachers’ conceptual understanding of particulate nature of matter through three-tier diagnostic test. International Journal of Education in Mathematics, Science and Technology, 5(3), 221-221.
Banawi, A., Sopandi, W., Kadarohman, A., & Solehuddin, M. (2019). Prospective primary school teachers’ conception change on states of matter and their changes through predict-observe-explain strategy. International Journal of Instruction, 12(3), 359-374. https://doi.org/10.29333/iji.2019.12322a
Barak, M., & Dori, Y. J. (2004). Enhancing undergraduate students’ chemistry understanding through project-based learning in an it environment. Science Education, 8(1), 117-139.
Brígido, M., Bermejo, M. L., Conde, C., & Mellado, V. (2010). The emotions in teaching and learning nature sciences and physics/chemistry in pre-service primary teachers. US-China Education Review, 7(12), 25-32.
Buss, R. R. (2010). Efficacy for teaching primary science and mathematics compared to other content. School Science and Mathematics, 110(6), 290-297. http://dx.doi.org/10.1111/j.1949-8594.2010.00037.x
Canpolat, N., & Pınarbaşı, T. (2012). Kimya öğretmen adaylarının kaynama olayı ile ilgili anlayışları: Bir olgubilim çalışması [Prospective chemistry teachers’ understanding of boiling: a phenomenological study]. Erzincan University Journal of Education Faculty, 14(1), 81-96.
Coştu, B., Ayas, A., Açıkkar, E., & Çalık, M. (2007). Çözünürlük konusuyla ilgili kavramlar ne düzeyde anlaşılıyor? [At which level are concepts about solubility topic understood?]. Boğaziçi University Journal of Education, 24(2), 13-28.
Coştu, B., Ayas, A., Çalık, M., Ünal, S., & Karataş, F. Ö. (2015). Fen öğretmen adaylarının çözelti hazırlama ve laboratuvar malzemelerini kullanma yeterliliklerinin belirlenmesi [Determining preservice science teachers’ competences in preparing solutions and in use of laboratory tools]. Hacettepe University Journal of Education, 28(28), 65-72.
Council of Higher Education [CoHE]. (2018). Sınıf öğretmenliği lisans programı, yeni öğretmen yetiştirme lisans programları [Primary education undergraduate program, new teacher training undergraduate programs].
Creswell, J. W. (2012). Educational research: Planning, conducting, and evaluating quantitative and qualitative research. Educational Research Vol. 4 (pp. 232-260). Harlow, United Kingdom: Pearson Education Limited.
Demirbaş, M., Tanrıverdi, G., Altınışık D., & Şahintürk, Y. (2011). Fen bilgisi öğretmen adaylarının çözeltiler konusundaki kavram yanılgılarının giderilmesinde kavramsal değişim metinlerinin etkisi [The impact of conceptual change texts on the elimination of misconceptions of science teacher candidates about the subject of solution]. Sakarya University Journal of Education, 1(2), 52-69.
Demircioğlu, H., Demircioğlu, G., & Ayas, A. (2004). Sınıf öğretmeni adaylarının bazı temel kimya kavramlarını anlama düzeyleri ve karşılaşılan yanılgılar [Prospective science teachers' levels of understanding and misconceptions about some basic chemical concepts]. HAYEF: Journal of Education, 1, 29-49.
Derman, A., & Ebenezer, J. (2020). The effect of multiple representations of physical and chemical changes on the development of primary pre-service teachers cognitive structures. Research in Science Education, 50, 1575–1601. https://doi.org/10.1007/s11165-018-9744-5
Donovan, M. S., & Bransford, J. D. (Eds.). (2005). How students learn: History, mathematics, and science in the classroom. Washington, DC: National Academies Press.
Duschl, R. A., Schweingruber, H. A., & Shouse, A. W. (Eds.). (2007). Taking science to school: Learning and teaching science in grades K-8. Washington, DC: National Academies Press.
Eilks, I., & Hofstein, A. (Eds.). (2015). Relevant chemistry education: From theory to practice. Rotterdam: Sense Publishers.
Gheith, E. M., & Aljaberi, N. M. (2015). Pre-service classroom teachers’ attitudes toward graphs and their ability to read and interpret them. International Journal of Humanities and Social Science, 5(7), 113-124.
Glazer, N. (2011). Challenges with graph interpretation: a review of the literature. Studies in Science Education, 47(2), 183-210.
Håland, B. (2010). Student-teacher conceptions of matter and substances – evaporation and dew formation. Nordina, 6(2), 109–124.
Härmälä-Braskén, A.-S., Hemmi, K., & Kurtén, B. (2020). Misconceptions in chemistry among Finnish prospective primary school teachers-a long-term study. International Journal of Science Education, 42(9), 1447-1464.
Head, M. L., Yoder, K., Genton, E., & Sumperl, J. (2017). A quantitative method to determine pre-service chemistry teachers' perceptions of chemical representations. Chemistry Education Research and Practice, 18(4), 825-840.
Herga, N. R., Čagran, B., & Dinevski, D. (2016). Virtual laboratory in the role of dynamic visualisation for better understanding of chemistry in primary school. Eurasia Journal of Mathematics, Science & Technology Education, 12(3), 593-608.
Hermita, N., Alpusari, M., Noviana, E., Putra, Z. H., Islami, N., Basori, H., Suhandi, A., & Samsudin, A. (2020). Improving prospective primary school teachers' mental models through implementation of CDOI supported by multimode visualization. Universal Journal of Educational Research, 8(2), 460-467. https://doi.org/10.13189/ujer.2020.080217
Januik, R. M., & Mazur, P. (2010). Poland. In B. Risch (Ed.), Teaching chemistry around the world (pp. 291-309). Germany: Hubert &Co.
Johnstone, A. H. (2000). Teaching of chemistry-logical or psychological? Chemistry Education Research and Practice, 1(1), 9–15.
Johnstone, A. H. (2010). You can’t get there from here. Journal of Chemical Education, 87(1), 22–29. https://dx.doi.org/10.1021/ed800026d
Juriševič, M., Glažar, S. A., Razdevšek Pučko, C., & Devetak, I. (2008). Intrinsic motivation of pre-service primary school teachers for learning chemistry in relation to their academic achievement. International Journal of Science Education, 30(1), 87–107. https://dx.doi.org/10.1080/09500690601148517
Kabapinar, F., Leach, J., & Scott, P. (2004). The design and evaluation of a teaching-learning sequence addressing the solubility concept with Turkish secondary school students. International Journal of Science Education, 26(5), 635—65.
Kelly, R. M., & Jones, L. L. (2008). Investigating students' ability to transfer ideas learned from molecular animations of the dissolution process. Journal of Chemical Education, 85(2), 303.
Kelly, R. M., Akaygün, S., Hansen, S. J. R., & Villalta-Cerdas, A. (2017). The effect that comparing molecular animations of varying accuracy has on students' submicroscopic explanations. Chemistry Education Research and Practice, 18(4), 582–600.
Knowles, M., Holton, E. F., & Swanson, R. A. (2005). The adult learner: the definitive classic in adult education and human resource development (6th ed.). Amsterdam: Elsevier.
Kozma, R. (2003). The material features of multiple representations and their cognitive and social affordances for science understanding. Learning and instruction, 13(2), 205-226.
Kurt, S., & Birinci Konur, K. (2017). Pre-service primary teachers’ impressions towards chemistry experiments based on constructivist laboratory approach. Inonu University Journal of the Faculty of Education, 18(3), 145-161. http://dx.doi.org/10.17679/inuefd.296545
Lemma, A. (2013). A diagnostic assessment of eight grade students’ and their teachers’ misconceptions about basic chemical concepts. African Journal of Chemical Education, 3(1), 39–59.
Lindawati, L., Wardani, S., & Sumarti, S. S. (2019). Development of inquiry materials based on chemical representation to improve students’ critical thinking ability. Journal of Innovative Science Education, 8(3), 332-343. https://doi.org/10.15294/JISE.V8I1.31082
Mansfield, C. F., & Woods-McConney, A. (2012). “I didn’t always perceive myself as a science person”: Examining efficacy for primary science teaching. Australian Journal of Teacher Education, 37(10),37-52.
Merino, C., & Sanmarti, N. (2008). How young children model chemical change. Chemistry Education Research and Practice, 9(3), 196–207.
Merriam, S. B. (1988). Case study research in education: a qualitative approach. San Francisco: Jossey-Bass.
Miles, M. B., & Huberman, A. M. (1994). Qualitative data analysis. United States of America Printed.
Nandiyanto, A. B. D., Asyahidda, F. N., Danuwijaya, A. A., Abdullah, A. G., Amelia, N., Hudha, M. N., & Aziz, M. (2018). Teaching “nanotechnology” for elementary students with deaf and hard of hearing. Journal of Engineering Science and Technology, 13(5), 1352-1363.
Okumuş, S., Öztürk, B., Doymuş, K., & Alyar, M. (2014). Maddenin tanecikli yapısının mikro ve makro boyutta anlaşılmasının sağlanması [Aiding comprehension of the particulate of matter at the micro and macro levels]. Journal of Educational Sciences Research, 4(1), 349-368.
Papageorgiou, G., Amariotakis, V., & Spiliotopoulou, V. (2017). Visual representations of microcosm in textbooks of chemistry: constructing a systemic network for their main conceptual framework. Chemistry Education Research and Practice, 18(4), 559-571. http://dx.doi.org/10.1039/C6RP00253F.
Papageorgiou, G., Kogianni, E., & Makris, N. (2007). Primary teachers’ views and descriptions regarding some science activities. Chemistry Education Research and Practice, 8(1), 52–60. https://dx.doi.org/10.1039/B6RP90019D
Pekdağ, B. (2010). Kimya öğreniminde alternatif yollar: animasyon, simülasyon, video ve multimedya ile öğrenme [Alternative methods in learning chemistry: learning with animation, simulation, video and multimedia]. Journal of Turkish Science Education Volume 7(2), 79-110.
Pine, K., Messer, D., & St. John, K. (2001). Children’s misconceptions in primary science: A survey of teachers’ views. Research in Science and Technological Education, 19(1), 79–96. https://dx.doi.org/ 10.1080/02635140120046240.
Petersen, J. E., & Treagust, D. F. (2014). School and university partnerships: The role of teacher education institutions and primary schools in the development of pre-service teachers' science teaching efficacy. Australian Journal of Teacher Education, 39(9), 153-167. http://dx.doi.org/10.14221/ajte.2014v39n9.2.
Polifka, J. (2021). Investigating introductory science students’ knowledge of multiple representations using technology. (Doctoral Dissertation). Retrieved from https://www.proquest.com/pqdtglobal/docview/2572566476/previewPDF/DB243A053C284C6BPQ/1?accountid=10527.
Sanchez, J. M. P. (2021). Understanding of kinetic molecular theory of gases in three modes of representation among tenth-grade students in chemistry. International Journal of Learning, Teaching and Educational Research, 20(1), 48-63. https://doi.org/10.26803/ijlter.20.1.3
Schulte, P. L. (2001). Pre-service elementary teachers' alternative conceptions in science and attitudes toward teaching science. (Doctoral Dissertation). Retrieved from https://www.proquest.com/docview/304703069
Sopandi, W., Kadarohman, A., Rosbiono, M., Latip, A., & Sukardi, R. R. (2018). The courseware of discontinuous nature of matter in teaching the states of matter and their changes. International Journal of Instruction, 11(1), 61–76. https://dx.doi.org/10.12973/iji.2018.1115a
Taber, K. S. (2009). Learning at the symbolic level. In J. K. Gilbert & D. Treagust (Eds.), Multiple representations in chemical education (pp. 75–105). Springer, Dordrecht.
Tarkın Çelikkıran, A., & Gökçe, C. (2019). Kimya öğretmen adaylarının çözünürlük konusuna ilişkin submikroskobik seviyedeki anlama düzeylerinin çizimlerle belirlenmesi [Determination of preservice chemistry teachers’ understanding of solubility concept at submicroscopic level by drawings]. Pamukkale University Journal of Education, 46, 57-87. http://dx.doi.org/10.9779/pauefd.457845
Taşdemir, A., & Demirbaş, M. (2010). İlköğretim öğrencilerinin fen ve teknoloji dersinde gördükleri konulardaki kavramları günlük yaşamla ilişkilendirebilme düzeyleri [The level of correlation of concepts that primary students seen topics in science and technology class with daily life]. International Journal of Human Sciences, 7(1), 124-148.
Taylor, N., & Coll, R. K. (2002). Pre-service primary teachers' models of kinetic theory: an examination of three different cultural groups. Chemistry Education Research and Practice, 3(3), 293-315.
Tepe, O., & Akkuzu Güven, N. (2020). Genel kimya konularına ilişkin grafik okuma, yorumlama ve çizim becerileri testi geliştirme süreci: Geçerlilik ve güvenilirlik analizleri [The development process of graph reading, interpreting and drawing skills test concerning general chemistry subjects: validity and reliability analyses]. Western Anatolia Journal of Educational Sciences, 11(1), 23-43.
Treagust, D. F., Chandrasegaran, A. L., Crowley, J., Yung, B. H. W., Cheong, I. P. A, & Othman, J. (2010). Evaluating students’ understanding of kinetic particle theory concepts relating to the states of matter, changes of state and diffusion: a cross-national study. International Journal of Science and Mathematics Education, 8, 141-164.
Tsaparlis, G., Kolioulis, D., & Pappa, E. (2010). Lower-secondary introductory chemistry course: A novel approach based on science-education theories, with emphasis on the macroscopic approach, and the delayed meaningful teaching of the concepts of molecule and atom. Chemistry Education Research and Practice, 11(2), 107–117.
Uluçınar Sağır, Ş., Tekin, S., & Karamustafaoğlu, S. (2012). Sınıf öğretmeni adaylarının bazı kimya kavramlarını anlama düzeyleri [The levels of prospective elementary school teachers’ understanding of some chemistry concepts]. Journal of Dicle University Ziya Gökalp Faculty of Education, 19, 112-135.
Uludüz, Ş. M. (2017). Sınıf öğretmeni adaylarının fen okuryazarlık düzeyleri ile fen öğretimi öz yeterlik inançlarının karşılaştırılması [The comparison of science literacy levels of primary teacher candidates and science teaching self efficacy beliefs]. (Master thesis). Retrieved from https://tez.yok.gov.tr/UlusalTezMerkezi/
Valanides, N. (2000). Primary student teachers' understanding of the particulate nature of matter and its transformations during dissolving. Chemistry Education: Research and Practice in Europe, 1(2), 249- 262.
Wach, E., & Ward, R. (2013). Learning about qualitative document analysis. Retrieved from https://opendocs.ids.ac.uk/opendocs/handle/20.500.12413/2989
Wang, Z., Chi, S., Luo, M., Yanga, Y., & Huanga, M. (2017). Development of an instrument to evaluate high school students’ chemical symbol representation abilities. Chemical Education Research and Practice, 18(4), 875-892. https://doi.org/10.1039/C7RP00079K
Wu, H. K., & Puntambekar, S. (2012). Pedagogical affordances of multiple external representations in scientific processes. Journal of Science Education and Technology, 21(6), 754-767.
Ye, J., Lu, S., & Bi, H. (2019). The effects of microcomputer-based laboratories on students macro, micro, and symbolic representations when learning about net ionic reactions. Chemistry Education Research and Practice, 20(1), 288-301. http://dx.doi.org/10.1039/c8rp00165k
Yerrick, R. K., & Simons, T. (2017). The affordances of fiction for teaching chemistry. Science Education International, 28(3), 232-243.
Yıldırım, A., & Şimşek, H. (2013). Sosyal bilimlerde nitel araştırma yöntemleri [Qualitative research methods in the social sciences]. Ankara: Seçkin Kitabevi.
DOI: https://doi.org/10.17509/jsl.v5i1.34772
Refbacks
- There are currently no refbacks.
Copyright (c) 2022 Melis Arzu Uyulgan, Nalan Akkuzu Güven

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.


Jl. Dr. Setiabudhi 229 Bandung 40154, West Java, Indonesia










